|
8/18/2023 0 Comments Chaperonincoli.Ĭhaperone Chaperonin Electron microscopy Equatorial split GroEL GroES Hsp60 Nanocomposite Protein folding Self-assembly. coli and its GroEL-GroES complex do not always receive in standard literature because the biochemical features of chaperonins derived from others special, such as mammals, are not always the same as those confirmed using GroEL-GroES derived from E. In parallel, a guideline is presented that supports the recognition that the E. In this article, an overview is presented on GroEL and the GroEL-GroES complex, with emphasis on their morphological variations, and some potential applications to the fabrication of nanocomposites using GroEL as a nano-block. 
A lot of knowledge on chaperonins has been accumulating as if expanding as ripples spread around the GroEL-GroES from Escherichia coli. There have been debates as to which complex is critical to the productive protein folding mediated by the GroEL-GroES complex, and how GroES coordinates with GroEL in the chaperonin reaction cycle in association with regulation by adenine nucleotides and through the interplay of substrate proteins. In the GroEL-GroES complex, a single heptameric GroEL ring binds one GroES ring in the presence of ATP/ADP, in this vein, the double ring GroEL tetradecamer is present in two distinct types of GroEL-GroES complexes: asymmetric 1:1 "bullet"-shaped GroEL:GroES and symmetric 1:2 "football" (American football)-shaped GroEL:GroES 2. The temperature-sensitive mutations in Hsp10 map to a domain (residues 25-40) that corresponds to a previously identified mobile loop region of bacterial GroES and result in a reduced binding affinity of hsp10 for the chaperonin at the non-permissive temperature.Chaperonin is categorized as a molecular chaperone and mediates the formation of the native conformation of proteins by first preventing folding during synthesis or membrane translocation and subsequently by mediating the step-wise ATP-dependent release that result in proper folding. The folding of the precursor of cytosolic dihydrofolate reductase (DHFR), imported into mitochondria as a fusion protein, is apparently independent of Hsp10 function consistent with observations made for the chaperonin-mediated folding of DHFR in vitro. Hsp10 is required for the folding and assembly of proteins imported into the matrix compartment, and is involved in the sorting of certain proteins, such as the Rieske Fe/S protein, passing through the matrix en route to the intermembrane space. 56 Type I chaperonins, in the evolutionarily connected bacterial cytoplasm, mitochondrial matrix, and. Our results identify Hsp10 as an essential component of the mitochondrial protein folding apparatus, participating in various aspects of Hsp60 function. The eukaryotic group II chaperonin TRiC/CCT assists the folding of 10 of cytosolic proteins including many key structural and regulatory proteins. Chaperonin rings alternate between two major states, a binding-active one, where an open ring binds a non-native protein in its central cavity, and a folding-active one, where the bound protein is released into a now-encapsulated cavity. The GroEL-induced unfolding of its substrate protein is only partial, requires the complete chaperonin system, and is accomplished within the 13 seconds required for a single system turnover. The HSP10 gene was cloned and sequenced and temperature-sensitive lethal hsp10 mutants were generated. The ability of the GroEL chaperonin to unfold a protein trapped in a misfolded condition was detected and studied by hydrogen exchange. To define the in vivo role of the co-chaperonin, we have used the genetic and biochemical potential of the yeast S. There has been great progress in understanding the structure and mechanism of action of the chaperonin family, exemplified by Escherichia coli GroEL. Mitochondria also contain a homologue of the cochaperonin GroES, called Hsp10, which is a functional regulator of the chaperonin. The molecular chaperones are a diverse set of protein families required for the correct folding, transport and degradation of other proteins in vivo.  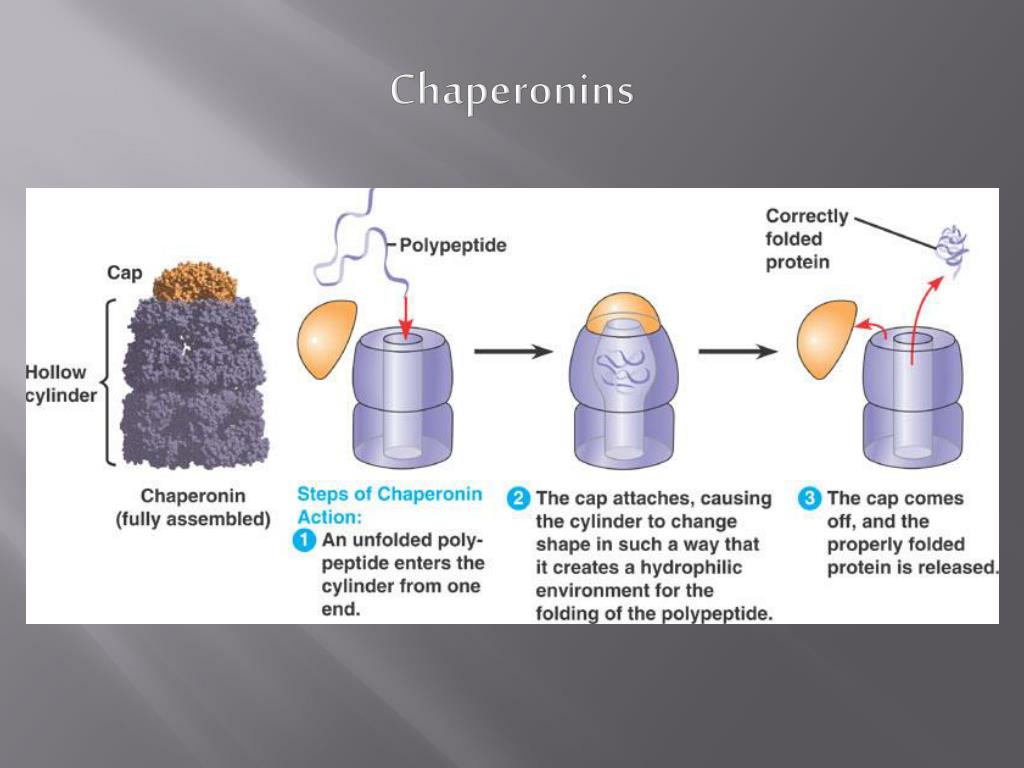
Protein folding in mitochondria is mediated by the chaperonin Hsp60, the homologue of E.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
